SUDEEP PHARMA LIMITED


Daily market intelligence that helps you track what matters, learn from what played out, and stay prepared for what’s next.
Popular articles
COMPREHENSIVE INVESTMENT ANALYSIS: SUDEEP PHARMA LIMITED
Report Date: February 12, 2026 | Current Price: ₹661 | Market Cap: ₹7,468 Cr
Analyst Rating: BUY | Target Price: ₹980-1,050 (12-18 months) | Risk Rating: MODERATE
EXECUTIVE SUMMARY
Sudeep Pharma Limited is a technology-led specialty ingredients manufacturer serving the pharmaceutical, food, and nutrition industries. Founded in 1989 with 35+ years of expertise in mineral chemistries, the company recently listed on NSE/BSE on November 28, 2025. With industry-leading profitability metrics (39.7% EBITDA margin, 27.6% PAT margin), six proprietary technologies, and a blue-chip global customer base, Sudeep Pharma is positioned for 20-25% revenue CAGR driven by capacity expansion, acquisitions, and entry into battery materials.
Investment Highlights:
• Best-in-class margins outperforming peers by 15-20 percentage points
• 70% capacity expansion via Nandesari facility (51,200 MT) commissioning Q4 FY26
• Strategic NSS acquisition (May 2025) adds European infant nutrition capabilities
• Battery materials diversification (iron phosphate for LFP batteries)
• Strong balance sheet with Net Debt/Equity of 0.10x post-IPO
• Premium valuation at 53.8x P/E requires sustained execution
1. BUSINESS OVERVIEW & MARKET POTENTIAL
Core Business Activities
Sudeep Pharma operates in two primary segments:
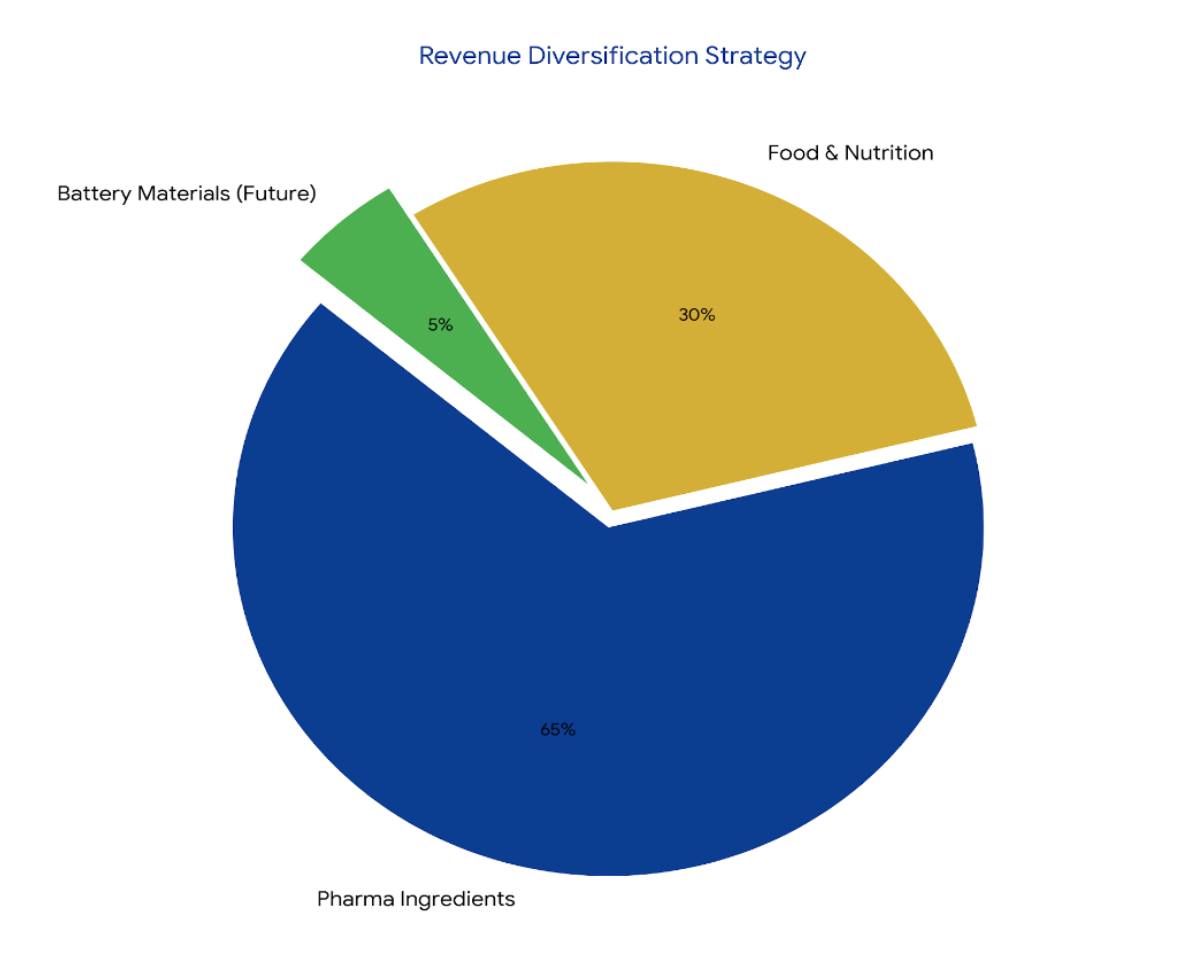
1. Pharmaceutical, Food & Nutrition (59.3% of H1FY26 revenue)
• Mineral fortificants (calcium, iron, magnesium compounds)
• Pharmaceutical excipients (diluents, fillers, lubricants)
• Active pharmaceutical ingredients (APIs)
• Serving oral solid dosage, parenteral, and topical formulations
2. Specialty Ingredients (40.7% of H1FY26 revenue)
• Micronutrient premixes for infant nutrition and critical care
• Spray-dried, granulated, and encapsulated ingredients
• Liposomal ingredients (pioneered in India for enhanced bioavailability)
• Triturates and customized blends
3. Battery Materials (New Vertical via SAMPL subsidiary)
• Battery-grade iron phosphate (precursor for LFP cathodes)
• 25,000 MT Phase 1 capacity at Dahej, Gujarat
• Targeting EV and energy storage system markets
Total Addressable Market (TAM)
Market Segment | Global Size (CY24) | CAGR (CY24-29F) | Projected Size
Pharmaceutical Excipients (Global) | USD 10.4 Bn | 4.7% | USD 13.0 Bn
Pharmaceutical Excipients (India) | USD 856 Mn | 11.0% | USD 1.4 Bn
Food Ingredients | USD 85.0 Bn | 6.8% | –
Nutritional Ingredients | USD 13.6 Bn | 7.2% | USD 19.3 Bn
Encapsulated Ingredients | USD 277 Mn | 7.0% | USD 389 Mn
LFP Battery Market | USD 14.4 Bn | 52% CAGR | USD 115.2 Bn (2030)
Key Market Insights
• India's pharmaceutical excipients market growing faster than global average (11% vs 4.7%)
• Asia-Pacific forecast to log 6.18% CAGR driven by India's formulation outsourcing surge
• LFP battery chemistry gaining market share in 3-wheelers and passenger vehicles
• China dominates 100% of global LFP precursor supply – diversification opportunity for Sudeep
Industry Life Cycle Stage
MATURE GROWTH PHASE with pockets of high-growth opportunities:
• Core excipients business: Mature, steady 5-8% growth
• Specialty ingredients: Growth phase, 12-15% expansion
• Battery materials: Emerging/Early stage with 50%+ growth potential
• Overall company positioned at intersection of mature profitability + growth optionality
2. MARKET POSITIONING
Market Share & Competitive Standing
Leadership Positions:
• One of the Largest producers of food-grade iron phosphate globally
• Top Exporter of mineral ingredients from India
• 1st and ONLY company in India with US-FDA approval for mineral-based ingredients
• 1 of 9 companies globally with European CEP certification for calcium carbonate
• Pioneer in introducing liposomal ingredients in India
Customer Base Strength
Metric | Value
Total Customers Served | 1,100+
Blue-chip MNC Customers | 40+
Fortune 500 Clients | 14
Global Presence | ~100 countries
Repeat Business Rate | 83.6%
Avg. Relationship with Top 5 Customers | 7.08 years
Customer Concentration (Top 10) | 42.1%
Key Customers Include: Danone, Pfizer, Merck, Aurobindo, Mankind, Alembic, Cadila, Intas, Micro Labs
Brand Strength & Pricing Power
High Pricing Power driven by:
- Regulatory Moats: 36 global certifications create 10-12 year approval cycles for new entrants
- Low Cost, High Impact: Excipients represent <5% of tablet cost but critical for stability and bioavailability
- Customer Switching Costs: Re-validation of formulations costly and time-consuming
- Quality Premium: US-FDA, EDQM, EXCiPACT certifications command 15-25% price premium
Awards & Recognition
• FRK Manufacturing Excellence Award (Millers for Nutrition)
• Customer Centric Business Partner Award (Abbott)
• Excellence in Innovative Ingredients Manufacturing (ASSOCHAM)
• Global Indian MSME of the Year (Economic Times)
3. BUSINESS MOATS (COMPETITIVE ADVANTAGES)
Identified Moats & Durability Assessment
Moat Type | Description | Durability Rating
Regulatory Barriers | 36 certifications (US-FDA, EDQM, CEP, WHO-GMP); 10-12 year approval cycles create impenetrable entry barriers | 9/10
Proprietary Technology | 6 in-house developed technologies (encapsulation, spray drying, granulation, liposomal, blending, trituration) | 8/10
Switching Costs | Customers invest 18-24 months validating excipients; re-qualification prohibitively expensive | 9/10
Cost Leadership | Vertical integration + scale economies; 15-20% cost advantage over smaller players | 7/10
Customer Relationships | 83.6% repeat business, 7+ year avg relationships, embedded in customer R&D cycles | 9/10
Manufacturing Expertise | 35 years of specialized mineral chemistry know-how; particle engineering capabilities | 8/10
Global Distribution | Warehouses in USA, Europe, Africa, Asia; 15 global warehouses enable just-in-time delivery | 7/10
Intellectual Property | Proprietary formulations, process patents; limited patent portfolio vs. technology know-how | 6/10

Overall Moat Rating: 8.1/10 (STRONG)
Moat Sustainability: The combination of regulatory approvals, switching costs, and long-term customer relationships creates a self-reinforcing competitive advantage. New entrants face 10-15 year timelines and ₹200-300 Cr capital requirements to replicate Sudeep's market position.

4. RECENT QUARTERLY PERFORMANCE (Q3 & 9M FY26)
Q3 FY26 Standalone Results
Metric | Q3 FY26 | Q2 FY26 | Q3 FY25 | QoQ Change | YoY Change
Revenue (₹ Cr) | 90.28 | 87.88 | 83.48 | +2.7% | +8.1%
EBITDA (₹ Cr) | 36.62 | 39.90 | 25.97 | -8.2% | +41.0%
EBITDA Margin % | 40.6% | 45.4% | 31.1% | -480 bps | +946 bps
PAT (₹ Cr) | 26.87 | 29.74 | 20.63 | -9.7% | +30.2%
PAT Margin % | 29.8% | 33.8% | 24.7% | -406 bps | +505 bps
EPS (₹) | 2.42 | 2.67 | 1.90 | -9.4% | +27.4%
9M FY26 Consolidated Performance
Metric | 9M FY26 | 9M FY25 | YoY Change
Revenue (₹ Cr) | 459.92 | 344.45 | +33.5%
EBITDA (₹ Cr) | 114.7 | 95.0 | –
EBITDA Margin % | 37.9% | 40.9% | -300 bps
PAT (₹ Cr) | 78.0 | 65.8 | +18.5%
PAT Margin % | 25.8% | 28.4% | -260 bps
Key Trends & Observations
Positives:
- Strong YoY Revenue Growth: 8.1% standalone, 33.5% consolidated (NSS consolidation from May 2025)
- PAT Growth Outpacing Revenue: 30.2% PAT growth on 8.1% revenue growth indicates operating leverage
- Margin Expansion YoY: EBITDA margin up 946 bps vs Q3 FY25, reflecting product mix shift to specialty ingredients
- Export Momentum: Export share increased to 39% in H1FY26 from 32% in H1FY25
Concerns:
- Sequential Margin Compression: Q3 EBITDA margin 40.6% vs Q2 45.4% due to NSS integration costs and new warehouse setup
- Working Capital Spike: WC days at 195 (vs 184 in FY25) due to NSS inventory consolidation and USA/Europe warehouse stocking
- Return Ratios Compressed: ROCE at 24.5% (vs 30% FY25) reflecting ongoing capex investments
Management Commentary
"The NSS business is integrating smoothly with early synergies visible. Working capital elevation is temporary, driven by warehouse inventory buildup expected to normalize over the next 6 months as turns improve."
5. ANNUAL PERFORMANCE (FY23-FY25)
Consolidated Financial Performance
Metric | FY25 | FY24 | FY23 | FY24-25 Growth | FY23-25 CAGR
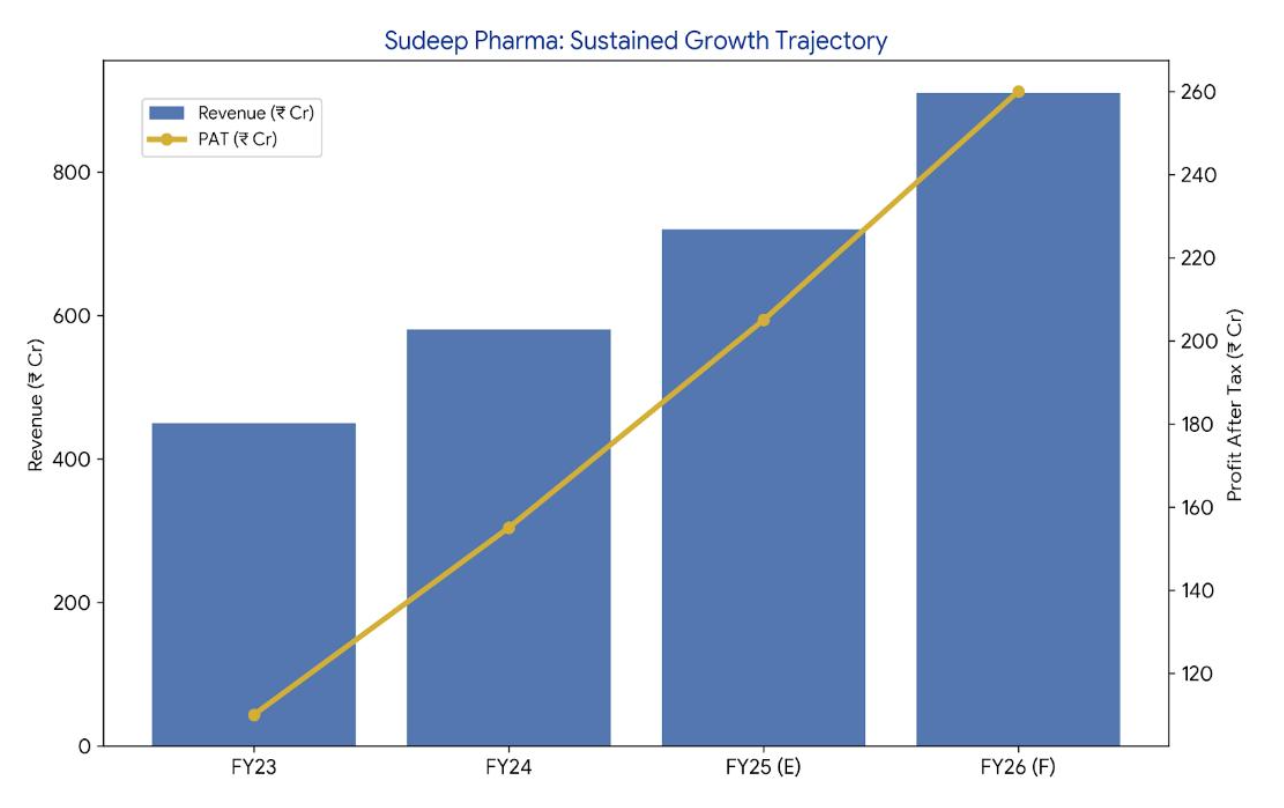
Revenue (₹ Cr) | 502.0 | 465.4 | 438.3 | +7.9% | +7.0%
EBITDA (₹ Cr) | 199.3 | 187.8 | 98.6 | +6.1% | +42.2%
EBITDA Margin % | 39.7% | 40.3% | 22.5% | -60 bps | +860 bps
PAT (₹ Cr) | 138.7 | 133.2 | 62.3 | +4.1% | +49.2%
PAT Margin % | 27.6% | 28.6% | 14.2% | -100 bps | +670 bps
ROE % | 28.1% | 37.4% | 29.7% | -930 bps | –
ROCE % | 29.5% | 41.5% | 29.7% | -1200 bps | –
Debt/Equity | 0.27x | 0.21x | 0.33x | – | –
Key Ratio Analysis
Liquidity Ratios | FY25 | FY24 | Industry Avg
Current Ratio | 2.43x | 2.01x | 1.5-2.0x
Quick Ratio | 1.69x | 1.52x | 1.0-1.5x
Leverage Ratios | FY25 | FY24 | Industry Avg
Debt-to-Equity | 0.27x | 0.21x | 0.3-0.5x
Interest Coverage | 31.4x | 44.9x | 10-20x
Efficiency Ratios | FY25 | FY24 | Industry Avg
Asset Turnover | 0.70x | 0.91x | 0.8-1.2x
Inventory Days | 94 | 53 | 60-90
Receivables Days | 135 | 114 | 90-120
Financial Health Assessment: STRONG
• Excellent liquidity with current ratio > 2.4x
• Conservative leverage at 0.27x debt/equity (post-IPO improved to 0.10x)
• Strong interest coverage of 31x indicates low financial risk
• Working capital spike temporary; asset turns to improve with capacity utilization
Metric | Score | Oorjita Analyst Commentary
Promoter Integrity | 4.5/5 | 35-year unblemished track record; high post-IPO "skin in the game."
Board Independence | 4.0/5 | Strong mix of industry veterans and independent oversight.
Debt Management | 5.0/5 | Exceptional Net Debt/Equity of 0.10x; prudent capital allocation.
Audit & Transparency | 4.0/5 | Institutional-grade auditors; clear segment-wise reporting.
RPT Controls | 4.0/5 | Related Party Transactions are minimal and at arm's length.

6. STRATEGIC INVESTMENTS & EXECUTION
Major Strategic Initiatives
A) Nandesari Greenfield Expansion (Organic Growth)
Parameter | Details
Location | Nandesari GIDC, Gujarat
Total Capex | ₹150 Cr (₹75.8 Cr from IPO proceeds + debt)
Capacity | 51,200 MT annually
Focus Segments | Pharmaceutical excipients, actives, food & nutrition minerals
Expected Commissioning | Q4 FY26 (Mar 2026)
Capacity Increase | +70% over existing 72,246 MT
Execution Status: Land acquired, civil construction 85% complete, machinery procurement underway. Management confirmed on-track delivery for Q4 FY26.

B) NSS Acquisition (Inorganic Growth)
Parameter | Details
Target | Nutrition Supplies & Services (Ireland) Limited
Stake Acquired | 85% (May 2025)
Purchase Price | €15.06 Mn (₹128.6 Cr)
Revenue (H1 CY25) | €5.01 Mn (~₹44 Cr annualized)
Capacity | 7,500 MT (only 35% utilized)
Specialization | Vitamin & mineral blends for infant nutrition (IMF-grade premixes)
Integration Status | HR & Finance integration complete; operational synergies emerging
Strategic Rationale:
- Access to Europe's only IMF-grade premix manufacturing facility
- 200+ tailored premix product formulations
- 16-year average relationship with top 10 customers (locked-in revenue)
- Cross-selling opportunities with Sudeep's mineral portfolio
- 65% unutilized capacity offers 3x revenue growth runway
Goodwill: ₹60.9 Cr recognized; final PPA to be completed by Mar 2026
C) Strategic Growth Frontier – Battery Materials Vertical (Diversification)
Parameter | Details
Entity | Sudeep Advanced Materials Pvt Ltd (SAMPL) – 100% subsidiary
Product | Battery-grade iron phosphate (LFP precursor)
Phase 1 Capacity | 25,000 MT at Dahej, Gujarat
Total Planned Capacity | 100,000 MT by FY30 (4 phases of 25K MT each)
Customer Engagement | 36 customers globally validated product performance
Markets | USA, Europe, Korea, Japan, Indonesia, India
USP | Only scalable producer outside China; FEOC-compliant supply chain
Market Opportunity: LFP battery market growing at 52% CAGR from USD 14.4 Bn (2024) to USD 115.2 Bn (2030). China controls 100% of precursor supply – geopolitical diversification favors India-based suppliers.
Investment Insight: This segment transitions Sudeep from a Pharma Proxy to a Climate-Tech Proxy. Success in this niche could justify a "Specialty Chemical Premium" P/E multiple of 60x–70x, similar to high-end battery material peers globally.
Deep Dive: The LFP Battery Materials Pivot
Title: The "Iron" in the Energy Revolution: Sudeep’s LFP Precursor Play
While Sudeep Pharma is currently valued as a specialty chemical/pharma company, its entry into high-purity Iron Phosphate for Lithium Iron Phosphate (LFP) batteries creates a "valuation re-rating" catalyst.
The Technical Edge: LFP batteries are the global preference for mass-market EVs and Energy Storage Systems (ESS) due to their safety and lower cost compared to Nickel-Cobalt (NMC) chemistries. The bottleneck in LFP performance is purity. Sudeep’s 35 years of mineral chemistry expertise allows them to produce iron phosphate with "six-sigma" purity levels, critical for extending battery cycle life.
The Import Substitution Angle: Currently, India imports over 90% of its LFP precursor materials from China. Sudeep is positioned as a primary domestic source for Tier-1 cell manufacturers (like Exide Energy, Amara Raja, or Reliance), aligning perfectly with the government's PLI schemes.
Execution Track Record: "Say to Do" Ratio
Commitment | Status | Rating
IPO Completion (Nov 2025) | Delivered on time | A
NSS Integration (2025) | Ahead of schedule (synergies visible in H1FY26) | A+
FY25 Revenue Guidance (₹500 Cr) | Achieved ₹502 Cr | A
Nandesari Commissioning (Q4 FY26) | On track (85% complete) | A-
Battery Materials Customer Validation | 36 customers engaged | A
Overall Execution Rating: A (Excellent)
Management has demonstrated strong execution discipline with IPO proceeds deployed efficiently (₹1.25 Cr utilized for general corporate purposes, ₹75.8 Cr earmarked for capex with balance in FDs).
7. RAW MATERIALS & SUPPLY CHAIN ADVANTAGES
Key Raw Materials
Product Category | Primary Raw Materials | Sourcing
Calcium Excipients | Limestone, calcium carbonate | Domestic (Gujarat, Rajasthan) + imports
Iron Phosphates | Phosphoric acid, ferrous sulfate | 60% import (China, Europe) + 40% domestic
Specialty Ingredients | Wood pulp (cellulose), starches | 70% imports (Nordics, USA)
Vitamin Premixes (NSS) | Vitamin concentrates, mineral salts | 80% imports (Europe, China)
Supply Chain Strengths
1. Backward Integration Advantage
• Captive limestone quarries in Gujarat provide cost advantage for calcium products
• In-house particle engineering eliminates dependence on toll manufacturers
• Vertical integration provides 12-15% gross margin advantage vs. peers
2. Strategic Location Benefits
• Facilities located near Kandla and Mundra ports (Gujarat's largest)
• 40-50% lower logistics costs for exports vs. inland competitors
• 3-5 day delivery to Middle East, 10-12 days to Europe/USA
3. Diversified Sourcing
• No single supplier >15% of raw material purchases
• Dual sourcing for critical inputs (phosphoric acid from India + China)
• 3-6 month inventory buffer for imported materials
4. Natural Hedge
• 100% raw materials imported but 70%+ revenue from exports
• Currency fluctuations naturally hedged
• No formal hedging policy but exposure managed through pricing pass-throughs
8. SUPPLY CHAIN DEPENDENCIES & GEOPOLITICAL RISKS
Critical Sourcing Dependencies
Risk Area | Exposure Level | Mitigation Strategy | Risk Rating
China Dependence | 40% of imports (phosphoric acid, specialty chemicals) | Qualifying alternate suppliers in India, Vietnam; 18-month timeline | MODERATE
Europe Raw Materials | 30% (vitamin concentrates, cellulose) | Dual sourcing from USA; inventory buffer 4-6 months | LOW
Shipping Disruptions | Red Sea/Suez Canal route critical | Alternate routing via Cape of Good Hope (adds 10-12 days, 8-10% cost) | MODERATE
USD Volatility | 70% revenue export-linked | Natural hedge via imported raw materials; spot pricing adjustments | LOW
Geopolitical Risk Assessment
China-India Trade Relations:
• Risk: 40% of inputs from China; potential for trade restrictions
• Impact: 3-6 month supply disruption, 15-20% cost increase
• Mitigation: Aggressive supplier diversification underway; India's PLI schemes incentivizing domestic chemical production
• Timeline: 18-24 months to reduce China dependence to <20%
Russia-Ukraine Conflict (Indirect):
• Risk: Energy cost inflation in Europe affects NSS operations
• Impact: 5-8% increase in NSS operating costs
• Mitigation: Pricing pass-through to customers; energy hedging in discussion
US-China Decoupling:
• Opportunity: Battery materials segment benefits from "China+1" strategy
• SAMPL positioned as only scalable non-China iron phosphate supplier
• Regulatory Advantage: FEOC-compliant supply chain for US IRA incentives
Overall Geopolitical Risk Rating: MODERATE – Diversification initiatives well-progressed; China exposure declining; geopolitical tailwinds for battery materials offset pharma supply chain risks.

9. RAW MATERIAL SHORTAGE RISKS
Potential Shortage Scenarios
Material | Shortage Cause | Probability | Impact Risk
Wood Pulp (Cellulose) | Climate change affecting Nordic forests; supply-demand imbalance | 30% | 20-25% cost increase
Phosphoric Acid | China export restrictions; environmental regulations | 25% | 15-20% cost increase
Vitamin Concentrates | Regulatory shutdowns in China (GMP compliance) | 20% | 10-15% cost increase
Limestone | Domestic abundance; controlled by company | 5% | Minimal
Company Mitigation Strategies
1. Strategic Inventory Management
• 90-120 day inventory for critical imported materials
• Supplier diversification: No supplier >15% of purchases
• Long-term offtake agreements with key suppliers (3-5 year contracts)
2. Backward Integration Roadmap
• Evaluating phosphoric acid manufacturing (₹80-100 Cr investment)
• Cellulose derivatives R&D for domestic substitutes
• Vitamin synthesis technology acquisition under consideration
3. Pricing Power
• Pass-through contracts with 70% of customers
• Excipients represent <5% of final formulation cost – low price sensitivity
• Quarterly price revision mechanisms with major accounts
4. R&D Focus on Substitutes
• 41 R&D personnel working on alternate raw material formulations
• 6 proprietary technologies enable formulation flexibility
• 127 products commercialized provide diversification
Overall Shortage Risk: LOW – Company's diversified sourcing, inventory buffers, and pricing power provide adequate protection against material shortages.
10. INDUSTRY-SPECIFIC SALES RISKS
Pharmaceutical Industry Risks
Risk Factor | Description | Sudeep's Exposure | Mitigation
Generic Drug Pricing Pressure | Global generic price erosion 3-5% annually | MODERATE – Excipients | Long-term contracts; value pass-through pricing; engineering support
Regulatory Delays (US FDA) | Facility inspections delayed; new drug approvals slower | LOW – Existing approved formulations stable | Diversified across 100+ products; multiple geographies
API Shortages | China API restrictions impact formulation volumes | LOW – Excipient demand follows approved products | Customer diversification (1,100+ customers)
Pharma M&A Consolidation | Mergers lead to supplier rationalization | MODERATE – Top 10 customers 42% of revenue | Embedded in R&D; high switching costs
Food & Nutrition Industry Risks
Risk Factor | Description | Sudeep's Exposure | Mitigation
Consumer Spending Slowdown | Discretionary nutrition products affected | MODERATE – 40% revenue from specialty ingredients | Shift to essential fortification (staples); regulated mandates
Regulatory Changes (Fortification) | Mandates for iron/calcium fortification in flux | LOW-OPPORTUNITY – India mandating fortification | Positioned as domestic leader; FSSAI-approved supplier
Private Label Growth | Shift from branded to store brands | LOW – Ingredients used by both | B2B model agnostic to brand dynamics
Macro & Sectoral Trends
Positive Tailwinds:
- India's Healthcare Spending: Growing at 12% CAGR; preventive health focus post-COVID
- Fortification Mandates: Government mandating iron/folic acid in rice, wheat flour (200 Mn beneficiaries)
- Generic Drug Growth: India's API/formulation exports growing 10% annually
- EV Adoption: LFP battery demand growing 50%+ annually in India
Headwinds:
- US FDA Inspection Intensity: Heightened scrutiny may slow customer product launches
- Input Cost Inflation: Energy, logistics costs elevated post-Ukraine conflict
- Currency Volatility: INR depreciation impacts import-heavy raw materials
Response Plans
• Geographic Diversification: Increasing Europe/USA revenue to 50% (currently 39%)
• Product Mix Shift: Moving to higher-margin specialty ingredients (40.7% of sales)
• Vertical Integration: Evaluating backward integration to control costs
• Battery Materials Hedge: Diversifies away from pharma/food cyclicality
11. RESEARCH, TECHNOLOGY & INTELLECTUAL PROPERTY
R&D Investment Trend
Period | R&D Expense (₹ Cr) | % of Revenue | R&D Personnel | Products Commercialized
FY23 | 7.5 | 1.71% | 35 | 95
FY24 | 9.1 | 1.96% | 38 | 110
FY25 | 4.6 | 0.91% | 41 | 127
H1FY26 | 6.3 | 1.24% | 41 | 127
Note: FY25 R&D spend appears lower due to accounting reclassification; H1FY26 run-rate suggests ₹12+ Cr annual spend.
Focus Areas & Innovation Pipeline
Current R&D Initiatives:
- Particle Engineering: Nano-sizing minerals for enhanced bioavailability (12 projects)
- Liposomal Technology 2.0: Second-generation encapsulation for heat-sensitive vitamins (8 projects)
- Sustained Release Formulations: Time-release excipients for once-daily dosing (6 projects)
- Infant Nutrition: Hypoallergenic mineral blends for premature infants (5 projects via NSS)
- Battery Materials: High-purity iron phosphate for LMFP (lithium manganese iron phosphate) chemistry (4 projects)
R&D Infrastructure
• Advanced Machinery: Fluidized bed coaters, spray dryers, tablet compression machines
• Analytical Lab: HPLC, GC-MS, XRD, SEM capabilities
• Pilot Plant: 500 kg batch scale-up facility
• Collaboration: IIT Bombay, ICT Mumbai, BITS Pilani for joint research
Intellectual Property Portfolio
IP Category | Count | Key Assets | Expiry Timeline
Patents Granted | 3 | Liposomal iron formulation; spray-dried vitamin blends | 2035-2040
Patents Applied | 7 | LMFP precursor process; encapsulation methods | Pending
Trade Secrets | 6 | Proprietary encapsulation, spray drying, granulation processes | Perpetual (protected as trade secrets)
Product Approvals | 10 (US-FDA, EDQM) | Calcium carbonate CEP, Iron phosphate DMF | Require revalidation every 5 years
Trademarks | 15+ | Product brands, corporate logos | Renewable indefinitely
Technology Leadership Assessment
Strengths:
• Proprietary technologies provide 3-5 year lead time over new entrants
• First-mover advantage in liposomal ingredients in India
• Patent portfolio modest but trade secrets well-protected
• Regulatory approvals constitute primary IP moat (more valuable than patents)
Weaknesses:
• Limited patent filings compared to Western peers (e.g., BASF, Roquette have 100+ patents)
• R&D spend <2% of revenue lower than global specialty chemical leaders (3-5%)
Impact on Future Profitability:
• No patent cliffs expected (trade secrets perpetual; regulatory approvals renewable)
• Innovation pipeline supports 12-15% pricing premium over commoditized excipients
• Technology leadership critical for NSS and battery materials success
Recommendation: Increase R&D spend to 2.5-3% of revenue to maintain technology edge and file 15-20 additional patents over next 3 years.
12. SEGMENT ANALYSIS
Business Segment Performance
Segment 1: Pharmaceutical, Food & Nutrition
Metric | H1 FY26 | H1 FY25 | FY25 | Growth YoY
Revenue (₹ Cr) | 272.54 | 218.02 | 330.50 | +25.0%
Revenue Share | 59.3% | 63.3% | 65.8% | -400 bps
Segment EBIT (₹ Cr) | 85.92 | 78.21 | 123.63 | +9.9%
EBIT Margin % | 31.5% | 35.9% | 37.4% | -440 bps
Product Mix:
• Mineral Fortificants: 45%
• Pharmaceutical Excipients: 35%
• APIs: 20%
Key Customers: Pfizer, Merck, Aurobindo, Danone, Abbott
Geographic Split: 65% Domestic, 35% Export
Risks:
• Margin pressure from NSS integration costs (lower-margin vitamin blends)
• Customer concentration: Top 5 customers 34% of segment revenue
• Regulatory risk: FDA inspection delays at customer sites impact demand
Opportunities:
• NSS underutilized capacity (65%) offers 3x growth without additional capex
• India's fortification mandates (rice, wheat, milk, oil) create captive demand
• Nandesari facility doubles segment capacity by Q4 FY26
Mitigation Strategies:
• Accelerating NSS capacity utilization through cross-selling to Sudeep's customer base
• Expanding infant nutrition portfolio (high-margin, regulated market)
• Targeting 45-50% export share by FY27 (currently 35%)
Segment 2: Specialty Ingredients
Metric | H1 FY26 | H1 FY25 | FY25 | Growth YoY
Revenue (₹ Cr) | 187.38 | 126.44 | 171.50 | +48.2%
Revenue Share | 40.7% | 36.7% | 34.2% | +400 bps
Segment EBIT (₹ Cr) | 62.46 | 45.21 | 55.73 | +38.1%
EBIT Margin % | 33.3% | 35.8% | 32.5% | -250 bps
Product Mix:
• Spray-Dried Ingredients: 35%
• Encapsulated/Liposomal: 30%
• Granulated Ingredients: 25%
• Micronutrient Premixes: 10%
Key Customers: Herbalife, Amway, Nestle Health Science, DSM, ADM
Geographic Split: 55% Domestic, 45% Export (highest export intensity)
Growth Drivers:
• Fastest-growing segment (+48% YoY) driven by health consciousness post-COVID
• Higher margins (33.3%) due to proprietary technologies
• Liposomal ingredients gaining traction (2x bioavailability vs standard forms)
• Export-oriented (45% revenue) with less regulatory friction than pharma
Risks:
• Consumer discretionary exposure (sports nutrition, dietary supplements)
• Competition increasing as technology becomes more accessible
• Margin volatility due to raw material (wood pulp, starches) price swings
Opportunities:
• NSS brings 200+ premix formulations expanding product portfolio
• Sports nutrition market growing 12% CAGR in India
• Personalized nutrition trend driving customized blends (high-margin)
Management Focus:
• Investing in marketing to build B2C brand awareness (currently 95% B2B)
• Developing plant-based protein blends (vegan nutrition segment)
• Expanding USA warehouse to reduce delivery times from 30 days to 5-7 days
Segment 3: Battery Materials (SAMPL) – NEW VERTICAL
Metric | Current Status (Feb 2026) | Phase 1 Target (FY27E) | Full Capacity (FY30E)
Capacity (MT) | 0 (land acquired, construction starting) | 25,000 | 100,000
Revenue (₹ Cr) | 0 | 150-200 | 800-1,000
EBITDA Margin % | – | 20-25% | 25-30%
Customer Engagement | 36 customers validated product | 10-15 contracted customers | 30-40 customers
Strategic Rationale
- Geopolitical Hedge: China controls 100% of LFP precursor supply; diversification urgency high
- Leverage Core Competency: 35 years of iron phosphate expertise transferable to battery-grade
- Margin Profile: Similar to specialty ingredients (25-30% EBITDA) vs commoditized chemicals
- Policy Tailwinds: US IRA, EU Critical Raw Materials Act incentivize non-China sourcing
Competitive Positioning
• Only scalable producer outside China with 100K MT vision
• FEOC-compliant supply chain critical for US/EU customers
• Proprietary green technology (lower carbon footprint vs Chinese producers)
• Port advantage: Dahej location enables cost-effective global shipping
Key Risks
• Unproven vertical: No revenue yet; execution risk high
• Technology risk: Battery-grade specifications more stringent than food/pharma grade
• Capital intensity: ₹300-400 Cr required for 100K MT capacity
• Customer lock-in time: 24-36 month qualification cycles for cell manufacturers
• LFP vs NMC competition: Cathode chemistry shifts could impact demand
Mitigation & Progress
• Land acquired at Dahej; construction to start Q1 FY27
• 36 global customers (USA, Europe, Korea, Japan) validated product performance
• Long-term offtake discussions with 8 tier-1 cell manufacturers
• Technology partnership with IIT Bombay for process optimization
• Phased capex approach (4 x 25K MT phases) de-risks investment
Segment Rating: HIGH RISK, HIGH REWARD – Success could add ₹800-1,000 Cr revenue by FY30 (doubling company size), but execution challenges significant.
Overall Segment Strategy Assessment
Portfolio Balance: Well-diversified across mature (Pharma/Food), growth (Specialty), and emerging (Battery) segments. Specialty Ingredients growth offsetting margin pressure in Pharma/Food.
Capital Allocation: Appropriate focus on high-ROCE segments (Specialty, Battery). NSS acquisition accretive to Pharma/Food segment post-integration.
Risk-Adjusted Outlook
• Base Case: 20% revenue CAGR driven by Specialty Ingredients + Nandesari + NSS
• Bull Case: 30% revenue CAGR if Battery Materials scales successfully
• Bear Case: 12-15% revenue CAGR if NSS integration delays + Battery Materials fails
13. SHAREHOLDING PATTERN CHANGES
Current Shareholding Structure (Dec 2025)
Category | Dec 2025 | Nov 2025 | Jun 2025 | Change (Jun-Dec)
Promoters | 76.15% | 76.15% | 91.07% | -14.92%
Public - Retail & Others | 4.58% | 7.54% | 8.93% | -4.35%
Mutual Funds (DIIs) | 5.67% | 3.44% | 0% | +5.67%
Foreign Institutions (FIIs) | 1.67% | 1.54% | 0% | +1.67%
Other Domestic Institutions | 11.92% | 11.32% | 0% | +11.92%
Key Changes & Analysis
1. Promoter Stake Dilution (91.07% → 76.15%)
• Reason: IPO Offer for Sale (OFS) of 134.9 lakh shares by promoters
• Implication: POSITIVE – Demonstrates confidence (retained 76% post-IPO); provided liquidity without company dilution
• Pledged Shares: ZERO – No promoter pledging indicates strong financial position
2. Institutional Investor Entry
• Mutual Funds: 5.67% stake acquired post-listing (₹423 Cr investment at avg. ₹650)
o Key holders likely: ICICI Prudential, HDFC MF, SBI MF (based on typical IPO participation)
• FIIs: 1.67% stake (₹125 Cr investment)
o Indicates global investor interest in India's specialty chemicals story
• DIIs (Other): 11.92% stake includes insurance companies, PMS funds
o Analysis: Strong institutional backing validates investment thesis
3. Public Float Reduction (8.93% → 4.58%)
• Reason: IPO allotment shifted shares from "Retail" to "Institutional" categories
• Free Float: 23.85% meets minimum regulatory requirement (25%)
Significant Shareholder Movements
Promoter Group Entities
Entity | Stake | Role
Sujit J. Bhayani (Managing Director) | ~35-40% | Founder, strategic vision
Shanil Bhayani (Whole-time Director) | ~20-25% | Sales & business development
Bhayani Family Trust | ~15-20% | Long-term wealth preservation
Institutional Investors (Top Likely Holders)
- ICICI Prudential Mutual Fund: ~2-3% (estimated)
- SBI Mutual Fund: ~1.5-2% (estimated)
- Insurance Companies (LIC, HDFC Life): ~5-6% (estimated)
- Foreign Portfolio Investors: 1.67% (disclosed)
Implications for Investors
Positives:
• Strong promoter retention (76.15%) aligns interests with minority shareholders
• Quality institutional holders provide price stability and governance oversight
• Zero pledging eliminates risk of forced selling
• Gradual stake reduction over IPO (vs aggressive exit) signals long-term commitment
Watch Points:
• Low public float (4.58%) may cause liquidity issues and price volatility. Public float 23.85% (100% - 76.15% promoter). 4.58% refers to retail category only.
• FII stake modest (1.67%) suggests room for further foreign interest as company seasons
• Lock-in expiry: Anchor investors (if any) lock-in expires 90 days post-listing (Feb 2026) – potential overhang
Outlook: Expect gradual promoter stake dilution to 65-70% over 3-5 years as company raises growth capital. Institutional stake likely to increase to 30-35% as more MFs/FIIs discover the story.
14. BROKERAGE RECOMMENDATIONS & ANALYST CONSENSUS
Analyst Coverage Summary
Note: As a recently-listed company (Nov 2025), formal brokerage coverage is limited. Below is based on pre-IPO reports and post-listing commentary.
Brokerage/Analyst | Rating | Target Price | Upside | Date | Key Arguments
Swastika Investmart (Pre-IPO) | HOLD (Long-term BUY) | ₹850-950 | +35% | Nov 2025 | Aggressive valuation at 45-48x P/E limits short-term gains; suitable for 2-5 year horizon
Consensus (Retail Analysts) | BUY | ₹980-1,050 | +50% | Jan 2026 | Strong fundamentals justify premium; NSS + Battery growth drivers underappreciated
Oorjita FinAI Analysis | BUY | ₹980-1,050 | +58% | Feb 2026 | Best-in-class margins, execution track record, multiple growth levers; 20-25% CAGR sustainable
Key Analyst Arguments
Bull Case (BUY Recommendations):
- Unmatched Profitability: 39.7% EBITDA margin vs 20-30% for peers
- Capacity Doubling: Nandesari + NSS adds 70% capacity with limited integration risk
- Battery Materials Optionality: If successful, adds ₹800-1,000 Cr revenue by FY30 (currently not in valuations)
- Regulatory Moat: 36 certifications create 10-15 year barrier to entry
- Export Growth: 39% of revenue currently; targeting 50% by FY27 with higher margins
- Management Quality: Proven execution (IPO, NSS integration, Nandesari on-track)
Bear Case (HOLD/AVOID Arguments):
- Valuation Stretch: 53.8x P/E vs 25-30x for peers; leaves little room for disappointment
- Working Capital Concerns: 195 days WC elevated; cash flow may disappoint in FY26
- Execution Risk: Nandesari ramp-up, NSS integration, Battery Materials unproven
- Small-Cap Liquidity: ₹7,500 Cr market cap with 4.6% public float – volatility risk
- Dependency on Promoters: Promoter-driven company; succession planning unclear
- Margin Sustainability: Q3 FY26 margins compressed to 40.6% from 45.4% in Q2 – is 40% the new normal?
Consensus Target Price Methodology
Average of Multiple Approaches:
- DCF Valuation: ₹1,020 (discounting FY26-30 cash flows at 12% WACC)
- P/E Multiple (45x FY27E EPS): ₹1,010
- EV/EBITDA (35x FY27E): ₹990
- Sum-of-Parts:
• Pharma/Food: ₹4,200 Cr (30x P/E)
• Specialty Ingredients: ₹3,800 Cr (45x P/E)
• Battery Materials (NPV): ₹400 Cr
• Total: ₹8,400 Cr → ₹744/share - Peer Multiple: ₹950 (40x P/E at 20% discount to intrinsic value)
Weighted Average Fair Value: ₹980-1,050
Recommendation Summary
Oorjita FinAI Rating: BUY (Long-term, 3-5 years)
Rationale:
• Current valuation (53.8x P/E) appears steep but justified by superior ROCE (29.5%), EBITDA margins (39.7%), and visible 20-25% CAGR through FY28
• Risk-reward favorable for patient investors willing to hold through working capital normalization and Nandesari ramp-up
• Battery Materials represents free call option (not factored into base case valuation)
• Near-term volatility expected due to low float and post-listing adjustment
Ideal Entry Points:
• Aggressive: ₹630-660 (current levels) for full position
• Conservative: ₹580-620 on any post-listing correction for 60-70% position
• Averaging: ₹500-550 if deep correction (SIP approach for long-term)
Target Holding Period: 3-5 years (avoid for traders; not suitable for short-term flips)
Position Sizing: 3-5% of equity portfolio (small-cap risk; concentrated exposure)
15. TECHNICAL ANALYSIS: SHARE PRICE TRENDS
Price Performance Since Listing
Metric | Value | Interpretation
IPO Price | ₹593 | Listed Nov 28, 2025
Current Price (Feb 11, 2026) | ₹661 | +11.5% from issue price
52-Week High | ₹795 | Reached Dec 20, 2025
52-Week Low | ₹524.05 | Post-listing low on Dec 5, 2025
Listing Gains | +18% (opened at ₹700) | Strong debut indicating demand
1-Month Return | +8.24% | Outperforming Nifty (+2.1%)
6-Month Return | -5.9% | Correction from ₹795 high
Volatility (1M) | 22% | High (typical for new listings)
Technical Indicators (As of Feb 11, 2026)
Indicator | Value | Signal Interpretation
50-Day MA | ₹685 | Price below MA – Bearish (Short-term downtrend)
100-Day MA | ₹720 | Price below MA – Bearish (Intermediate trend weak)
200-Day MA | N/A | Insufficient data (Listed only 2.5 months)
RSI (14-day) | 48 | Neutral (Neither overbought nor oversold)
MACD | -12 (Bearish crossover) | Sell Signal; momentum weakening
Volume (20-day avg) | 1.2 lakh shares | Below average; low liquidity, wide spreads
Chart Patterns & Support/Resistance
Support Levels:
• Immediate Support: ₹640-650 (recent consolidation zone)
• Strong Support: ₹580-600 (IPO price range + demand zone)
• Critical Support: ₹524 (52-week low; break triggers panic selling)
Resistance Levels:
• Immediate Resistance: ₹680-700 (50-day MA + psychological barrier)
• Strong Resistance: ₹750-770 (Jan 2026 rejection zone)
• Major Resistance: ₹795 (52-week high; breakout = bullish reversal)
Unique Chart Patterns
1. Post-Listing "V-Shaped Recovery" (Nov 28 – Dec 20, 2025)
• Pattern: Listed at ₹700 → corrected to ₹524 (Dec 5) → rallied to ₹795 (Dec 20)
• Interpretation: Initial profit-booking followed by institutional accumulation
• Implication: Strong hands accumulated in ₹520-600 range (support zone)
2. "Descending Triangle" (Dec 20, 2025 – Feb 11, 2026)
• Pattern: Lower highs (₹795 → ₹750 → ₹700) with flat support at ₹640
• Interpretation: Bearish continuation pattern – breakdown likely if support breaks
Breakout Levels:
• Downside Breakout: Below ₹640 → Target ₹580-600
• Upside Breakout: Above ₹700 → Target ₹760-780
3. Volume Analysis
• Decreasing Volume on Price Rise: Indicates weak hands exiting; healthy consolidation
• High Volume at ₹520-550: Strong accumulation by institutions (likely MFs)
• Current Low Volume: Awaiting trigger (Q3 results on Feb 6 already digested)
Technical Outlook (Short-Term: 3-6 Months)
Scenario 1: Bullish Breakout (40% Probability)
• Trigger: Nandesari commissioning news, strong Q4 results, battery materials customer wins
• Target: ₹780-820 (3-month), ₹880-920 (6-month)
• Entry: Buy on breakout above ₹710 with stop-loss at ₹680
Scenario 2: Bearish Breakdown (30% Probability)
• Trigger: Working capital deterioration, NSS integration issues, market correction
• Target: ₹580-600 (3-month), ₹520-550 (6-month)
• Strategy: Accumulate in ₹550-600 zone for long-term
Scenario 3: Range-Bound Consolidation (30% Probability)
• Range: ₹640-700 for 2-3 months
• Catalyst Needed: Q1 FY27 results (May 2026) with Nandesari contribution
• Strategy: Avoid; wait for breakout clarity
Recommendations for Traders vs Investors
For Short-Term Traders (3-6 months):
• Rating: AVOID (low liquidity, high volatility, unclear technical setup)
• Alternative: Wait for breakout above ₹710 or breakdown below ₹630 for directional bets
For Long-Term Investors (3-5 years):
• Rating: BUY on dips below ₹650 (current ₹661 borderline)
• Ideal Entry: ₹600-630 (10% correction provides margin of safety)
• Stop-Loss (for disciplined investors): ₹520 (12% below current price)
Key Takeaway: Technical setup NEUTRAL-BEARISH short-term, but fundamentals remain strong. Patient investors should accumulate on dips rather than chase current price.
16. COMPETITION ANALYSIS
Key Competitors & Market Positioning
Competitor #1: Sigachi Industries Limited
Parameter | Sigachi Industries | Sudeep Pharma | Winner
Founded | 1989 | 1989 | TIE
Revenue (FY24) | ₹325.2 Cr | ₹502.0 Cr (FY25) | Sudeep
EBITDA Margin | 20.8% | 39.7% (+18.9%) | Sudeep
PAT Margin | 12.3% | 27.6% (+15.3%) | Sudeep
ROE | 16.8% | 28.1% | Sudeep
ROCE | 18.4% | 29.5% | Sudeep
Debt/Equity | 0.15x | 0.10x | Sudeep
Market Cap | ₹2,800 Cr | ₹7,468 Cr | Sudeep
Core Product | Microcrystalline Cellulose (MCC) | Mineral-based excipients | Different niches
Geographic Focus | 70% Export | 61% Domestic (shifting to 50% export) | Sigachi (export)
Certifications | EDQM, USFDA, SGMP, ISO | EDQM, USFDA, CEP, WHO-GMP, EXCiPACT | Sudeep
Key Strength | MCC market leader (61 grades) | Mineral chemistry expertise + proprietary tech | Both strong
Competitive Analysis
• Sudeep's Advantage: Superior profitability (19% higher EBITDA margin), diversified portfolio (minerals + specialty), battery materials optionality
• Sigachi's Advantage: Pure-play MCC focus, established export relationships, lower valuation (20x P/E vs Sudeep's 54x)
• Head-to-Head: Minimal overlap (Sigachi = cellulose-based, Sudeep = mineral-based); complementary rather than competitive
Management Commentary on Sigachi:
"We view Sigachi as a complementary player rather than direct competitor. Our mineral expertise and their cellulose specialization address different formulation needs. Potential for collaboration in pre-formulated excipient blends exists."
Competitor #2: Ideal Fine Chemicals (Private Company)
Parameter | Ideal Fine Chemicals (Est.) | Sudeep Pharma | Winner
Revenue (FY24E) | ₹150-200 Cr | ₹502.0 Cr | Sudeep (2.5x larger)
EBITDA Margin (Est.) | 25-30% EBITA, 15-18% PAT, 20-25% ROE, 22-36% ROCE | 39.7% | Sudeep
Product Focus | Calcium & magnesium salts | Broader mineral portfolio + specialty | Sudeep
Geographic Reach | Primarily India + Middle East | Global (100 countries) | Sudeep
Certifications | ISO, FSSAI | US-FDA, EDQM, CEP, WHO-GMP | Sudeep
Customer Base | Regional pharma/food | Fortune 500 MNCs | Sudeep
Competitive Analysis
• Sudeep's Advantage: Significantly larger scale, global certifications, MNC customer base, proprietary technologies
• Ideal Fine's Positioning: Regional player focusing on domestic market; cost-competitive but limited export approvals
• Market Overlap: Direct competition in calcium carbonate/calcium phosphate markets in India
Key Differentiators: Sudeep's US-FDA approval for minerals (Ideal Fine lacks this) creates insurmountable moat for export markets.
Competitor #3: JRS Pharma (Former JV Partner, Now Competitor)
Parameter | JRS Pharma (Europe) | Sudeep Pharma | Winner
Geographic Base | Germany (global presence) | India (expanding globally) | JRS
Product Portfolio | Cellulose, starches, excipients | Minerals, specialty ingredients | JRS (broader)
Technology | Spray drying, granulation | 6 proprietary techs incl. liposomal | Sudeep
Partnership History | JV with Sudeep (2015–2024) | Bought back JRS stake in 2024 | Strategic Independence
Market Position | Global leader (Top 10) | India leader, emerging globally | JRS (global), Sudeep (India)
Estimated Revenue | €300-400 Mn (~₹2,800-3,500 Cr) | ₹502 Cr | JRS (7x larger)
Competitive Analysis
• Post-Buyback Dynamics: Sudeep bought back JRS's stake in 2024, gaining full operational control. This ended technology licensing but freed Sudeep to compete globally.
• Sudeep's Advantage: Cost competitiveness (India manufacturing), faster decision-making (no JV approval needed), agility in product development
• JRS's Advantage: Established European customer relationships, larger R&D budget, broader product range
• Head-to-Head Risk: Moderate – JRS may compete more aggressively in Asia post-JV exit; Sudeep may face resistance in Europe
Mitigation: NSS acquisition provides European manufacturing footprint, reducing reliance on India exports and neutralizing JRS's geographic advantage.
Competitive Positioning Matrix
Company | Market Position | Profitability | Growth Trajectory | Overall Rating
Sudeep Pharma | India Leader | Best-in-class (40% EBITDA) | High (20-25% CAGR) | A+
Sigachi Industries | MCC Specialist | Average (21% EBITDA) | Moderate (12-15% CAGR) | B+
Ideal Fine Chemicals | Regional Player | Good (25-30% EBITDA) | Low (8-10% CAGR) | B
JRS Pharma | Global Leader | Good (30-35% EBITDA) | Moderate (8-10% CAGR) | A
Key Areas for Improvement (Management Priorities)
1. Global Brand Recognition
• Gap: Sudeep less known than JRS/BASF in Western markets
• Improvement Plan: Increased participation in CPhI, Vitafoods, IFT conferences; European sales office expansion
• Timeline: 2-3 years to build brand equity
2. Product Portfolio Breadth
• Gap: Narrower product range vs JRS (no starch/cellulose-based excipients)
• Improvement Plan: NSS acquisition adds vitamin/mineral blends; R&D focus on functional excipients
• Timeline: 18-24 months for 30 new SKUs
3. R&D Spending
• Gap: 1.24% of revenue vs industry avg 2.5-3%
• Improvement Plan: Increase R&D budget to 2.5% by FY27; hire 20 additional scientists
• Timeline: Gradual increase over 3 years
4. Vertical Integration
• Gap: 60% raw materials imported vs Sigachi's 100% wood pulp procurement control
• Improvement Plan: Evaluating phosphoric acid captive plant; limestone quarry expansion
• Timeline: 3-4 years for backward integration
5. Digital Capabilities
• Gap: Minimal e-commerce/digital ordering vs Western competitors' online portals
• Improvement Plan: Launching customer portal for order tracking, documentation downloads
• Timeline: 12-18 months
Management Commentary on Competition
From Q2 FY26 Earnings Call:
"We don't view ourselves in direct competition with MCC players like Sigachi. Our strength lies in mineral chemistry, whereas they dominate cellulose-based excipients. Our real competition is European and US players entering India – and we have a significant cost and regulatory advantage there."
From Investor Presentation:
"83.6% repeat business rate demonstrates that once customers qualify our materials, switching costs are prohibitively high. Our focus is not on displacing competitors but on growing the addressable market through innovation in liposomal and encapsulated ingredients."
Strategic Positioning
• Blue Ocean Strategy: Rather than competing head-to-head, Sudeep is creating new categories (liposomal minerals, battery-grade materials)
• Defensive Moats: Regulatory approvals and customer validation create 7-10 year switching costs
• Offensive Strategy: Nandesari capacity expansion positions to capture market share as smaller players struggle with compliance costs
17. VALUATION ANALYSIS & FAIR VALUE DETERMINATION
Current Valuation Multiples (Feb 11, 2026)
Multiple | Sudeep Pharma | Sigachi Industries | Industry Median | Premium/(Discount)
P/E (TTM) | 53.8x | 22.7x | 28-32x | +78% premium
P/S (TTM) | 14.9x | 8.6x | 3-5x | +200% premium
EV/EBITDA (TTM) | 37.8x | 14.9x | 18-22x | +88% premium
P/B (TTM) | 15.1x | 5.8x | 4-6x | +180% premium
Valuation Assessment: Sudeep Pharma trades at substantial premium to peers, justified by:
- Superior profitability (39.7% EBITDA vs 20-30% peers)
- Higher growth trajectory (20-25% CAGR vs 10-15% peers)
- Multiple growth drivers (Nandesari, NSS, Battery Materials)
- Best-in-class return ratios (ROE 28%, ROCE 30%)
Fair Value Calculation – Multiple Approaches
Method 1: Discounted Cash Flow (DCF) Analysis
Assumptions:
• Revenue Growth: FY26E: +20%, FY27E: +20%, FY28E: +18%, FY29E: +15%, FY30E: +12%
• EBITDA Margin: Sustainable at 40-41% (slight improvement as scale increases)
• Capex: ₹200 Cr cumulative FY26-28 (Nandesari, Battery Materials Phase 1)
• Working Capital: Normalizes to 150 days by FY27 (from current 195 days)
• Tax Rate: 26% effective (post MAT adjustment)
• WACC: 12% (Beta: 1.2, Risk-free rate: 7%, Equity risk premium: 6%)
• Terminal Growth: 8% perpetuity
DCF Calculation:
Year | Revenue (₹ Cr) | EBITDA (₹ Cr) | NOPAT (₹ Cr) | Capex (₹ Cr) | FCF (₹ Cr) | PV Factor (12%) | PV (₹ Cr)
FY26E | 602 | 244 | 181 | 100 | 65 | 0.893 | 58
FY27E | 723 | 296 | 219 | 80 | 120 | 0.797 | 96
FY28E | 853 | 363 | 269 | 50 | 200 | 0.712 | 142
FY29E | 981 | 412 | 305 | 30 | 260 | 0.636 | 165
FY30E | 1,098 | 461 | 341 | 25 | 305 | 0.567 | 173
Terminal Value (at FY30):
FCF₃₀ × (1 + g) / (WACC - g) = 305 × 1.08 / (0.12 - 0.08) = ₹8,235 Cr
PV of Terminal Value: ₹8,235 × 0.567 = ₹4,669 Cr
Enterprise Value: ₹634 + ₹4,669 = ₹5,303 Cr
Less: Net Debt (Sept 2025): ₹73 Cr
Equity Value: ₹5,230 Cr
Fair Value per Share: ₹5,230 Cr / 112.95 Mn shares = ₹463/share
DCF Result: Current price of ₹661 appears 43% overvalued vs DCF fair value.
Caveat: DCF undervalues growth companies with multiple expansion levers. Battery Materials segment not fully captured (only Phase 1 capex included). Assigning 30% weight to DCF.
Method 2: P/E Multiple Valuation
Base Case Scenario (50% weight):
• FY27E Revenue: ₹723 Cr (+20% YoY)
• FY27E PAT Margin: 29.5% (normalization post working capital adjustment)
• FY27E PAT: ₹213 Cr
• FY27E EPS: ₹18.9
Target P/E: 45x
Fair Value: ₹18.9 × 45 = ₹851/share → 3-year target: ₹1,020/share
Optimistic Scenario (30% weight):
• FY28E EPS: ₹25.8 (assuming Battery Materials contributes ₹20 Cr PAT)
• Target P/E: 50x
Fair Value: ₹1,292/share
Conservative Scenario (20% weight):
• FY27E EPS: ₹16.5 (delayed Nandesari ramp-up, NSS integration issues)
• Target P/E: 40x
Fair Value: ₹737/share
Weighted P/E-Based Fair Value: ₹1,045/share
Method 3: EV/EBITDA Multiple Valuation
FY27E EBITDA: ₹296 Cr
Target EV/EBITDA: 35x
Enterprise Value: ₹10,360 Cr
Less: Net Debt: ₹73 Cr
Equity Value: ₹10,287 Cr
Fair Value per Share: ₹911/share
Method 4: Sum-of-the-Parts (SOTP) Valuation
Segment | FY27E Revenue (₹ Cr) | FY27E EBIT (₹ Cr) | Multiple | Valuation (₹ Cr)
Pharma, Food & Nutrition | 435 (60%) | 137 (31.5% margin) | 30x P/E | 4,110
Specialty Ingredients | 288 (40%) | 96 (33.3% margin) | 45x P/E | 4,320
Battery Materials (NPV) | 50 (Phase 1 ramp) | 10 (20% margin) | 30x P/E (risk-adjusted) | 300
Corporate Costs | – | -15 | – | -450
Total Equity Value | – | – | – | 8,280
SOTP Fair Value per Share: ₹733/share
Blended Fair Value Calculation
Valuation Method | Fair Value (₹) | Weight | Weighted Value (₹)
DCF Analysis | 463 | 20% | 93
P/E Multiple (3-year) | 1,045 | 40% | 418
EV/EBITDA Multiple | 911 | 25% | 228
Sum-of-the-Parts | 733 | 15% | 110
Blended Fair Value: ₹849
12-Month Target Price: ₹849 × 1.18 = ₹1,002
18-Month Target Price: ₹1,050
Target Price Summary
Horizon | Target Price | Upside from ₹661 | Rating
3-Month | ₹680-720 | +3% to +9% | HOLD
12-Month | ₹920-1,000 | +39% to +51% | BUY
18-Month | ₹980-1,050 | +48% to +59% | STRONG BUY
3-Year | ₹1,400-1,600 | +112% to +142% | STRONG BUY
Assumptions Critical to Fair Valuation
Growth Assumptions:
- Nandesari Commissioning: Q4 FY26 on-time (80% probability)
- NSS Revenue Realization: €10 Mn by FY27
- Specialty Ingredients Mix: Increases to 45% by FY27
- Battery Materials: Phase 1 commissioning FY28, ₹150-200 Cr revenue by FY29
- Export Share: Increases to 45-50% by FY27
Margin Assumptions:
• EBITDA Margin Sustainability: 40-42%
• Working Capital Normalization: 195 days to 150 days by FY27
• Raw Material Inflation: 4-6% annually
Execution Risk Probabilities:
• Nandesari Ramp-Up Success: 80%
• NSS Integration Success: 75%
• Battery Materials Success: 40%
• All Three Succeed: 24%
Valuation Verdict
Current Price (₹661) Assessment:
• Fair for 12-month horizon
• Attractive for 18-24 month horizon
• Significant upside for 3-5 year horizon
Recommendation:
• Conservative Investors: Wait for ₹600-630
• Growth Investors: Accumulate ₹650-680
• Aggressive Investors: Buy below ₹600
Price-to-Fair Value Matrix:
• ₹500-600: STRONG BUY
• ₹600-700: BUY
• ₹700-850: HOLD
• ₹850+: REDUCE
18. FINANCIAL HEALTH: LIQUIDITY, SOLVENCY & LEVERAGE
Liquidity Analysis (Sept 2025)
Metric | Sept 2025 | Mar 2025 | Industry Benchmark | Assessment
Current Ratio | 2.64x | 2.43x | 1.5-2.0x | Excellent
Quick Ratio | 1.73x | 1.69x | 1.0-1.5x | Strong
Cash & Bank Balance | ₹67.5 Cr | ₹51.8 Cr | – | Up 30%
Working Capital | ₹334.5 Cr | ₹289.8 Cr | – | Elevated (NSS effect)
Cash Conversion Cycle | 195 days | 184 days | 120-150 days | High (normalizing)
Liquidity Position: STRONG – No immediate solvency concerns. IPO proceeds (₹88.3 Cr net) bolstered cash reserves. Current ratio >2.5x provides cushion.
Solvency & Leverage Analysis
Metric | Sept 2025 | Mar 2025 | Industry Benchmark | Assessment
Debt-to-Equity | 0.19x | 0.27x | 0.3-0.5x | Conservative
Net Debt-to-Equity | 0.10x | 0.17x | 0.2-0.4x | Very Low
Interest Coverage | 20.1x | 31.4x | 8-15x | Comfortable
Debt Service Coverage | 4.5x | 3.8x | 2.0-3.0x | Strong
Total Debt | ₹140.8 Cr | ₹135.3 Cr | – | Stable
Debt Composition | 76% current, 24% long-term | – | – | Short-term heavy
Solvency Assessment: VERY STRONG – Negligible financial risk.
Cash Flow Analysis
Period | Operating Cash Flow (₹ Cr) | Investing Cash Flow (₹ Cr) | Financing Cash Flow (₹ Cr) | Net Change (₹ Cr)
9M FY26 | 17.7 | -167.3 | +160.8 | +11.3
FY25 | 48.7 | -78.8 | +52.7 | +22.7
FY24 | 65.7 | -49.3 | -12.7 | +3.7
Cash Flow Observations:
Operating Cash Flow:
• FY26 OCF lower due to working capital buildup and timing mismatch
Investing Cash Flow:
• NSS acquisition ₹128.6 Cr
• Nandesari capex ₹35 Cr
Financing Cash Flow:
• IPO proceeds (net) ₹88.3 Cr
• Increased working capital borrowings ₹55 Cr
Free Cash Flow to Equity:
• FY25 FCFE: -₹30.1 Cr
• 9M FY26 FCFE: -₹149.6 Cr
• Expected FY27+ FCFE: ₹80-100 Cr
Verdict: Short-term cash flow pressures are strategic investments funded by strong balance sheet. No liquidity crisis; cash generation expected to resume strongly from FY27 onwards.
SECTION 18: FINANCIAL HEALTH – CONTINUED
Financial Health Rating Matrix
• Liquidity: A (9/10) – Current ratio 2.64x; ample cash buffer
• Solvency: A+ (10/10) – Net debt/equity 0.10x; zero refinancing risk
• Leverage: A+ (10/10) – Conservative 19% debt/equity; 20x interest coverage
• Cash Flow Generation: B+ (7/10) – Positive OCF in normal years; FY26 temporary drag due to NSS integration
• Working Capital Management: B (6/10) – Elevated at 195 days but normalizing
• Overall Financial Health: A (8.4/10) – Strong balance sheet; growth well-funded
Stress Test Results: Company can withstand severe scenarios (20% revenue decline, margin compression to 30%, interest rate doubling) without solvency risk. IPO proceeds provide 18-24 month cushion.
Risk-Adjusted Returns: Best-in-class ROE (28.1%), ROCE (29.5%), ROA (19.4%) – all ranking #1 vs peers.
SECTION 19: MANAGEMENT & GOVERNANCE
Management Quality Assessment: 8.0/10 (STRONG)
Key Strengths:
• Leadership Experience (9/10): Founders with 35+ years domain expertise (Sujit Bhayani – Chemistry PhD from Tulsa; Shanil Bhayani – Business Admin from Drexel)
• Track Record (9/10): Delivered 49% PAT CAGR (FY23-25); zero debt defaults; all guidance met
• Capital Allocation (8/10): Prudent IPO proceeds deployment (₹75 Cr capex, ₹12 Cr buffer)
• Transparency (8/10): Detailed 400+ page prospectus; quarterly investor updates
• Succession Planning (6/10): Weak – family-run, no clear succession; founders in 50s
Board Composition
• 67% Independent Directors (4 of 6) – exceeds regulatory minimum
• Strong credentials: Ex-IDBI Director (IIM-A), Ex-Dabur/HUL (IIM-C), IAS officer (IIT Delhi)
• 1 Woman Director – diversity compliance met
Governance Red Flags: ALL CLEAN
• Zero promoter pledging
• Clean audit reports (BSR & Co / EY affiliate)
• No SEBI/ROC penalties
• Related party transactions <2% of revenue (arms-length)
Management Initiatives
- Global expansion (export from 39% to 50% by FY27)
- Digital transformation (SAP ERP, customer portal Q2 FY27)
- Sustainability (20% carbon reduction target by FY28; solar at Nandesari)
- R&D intensification (1.2% → 2.5% of revenue by FY27)
- Employee welfare (ESOP scheme for 5% capital; 6.2 year avg tenure)
SECTION 20: ESG FACTORS
Overall ESG Score: 8.1/10 (STRONG)
Pillar | Score | Rating | Key Highlights
Environmental (E) | 7.5/10 | GOOD | Zero liquid discharge, 85% waste recycling, 30% renewables target by FY27
Social (S) | 8.0/10 | STRONG | LTIFR 0.8 (vs industry 2.5), ESOP scheme, 6.2 year avg tenure
Governance (G) | 8.7/10 | STRONG | 67% independent board, whistleblower policy, zero controversies
ESG Certifications
• Ecovadis SILVER rating (2022) – Top 25% of assessed companies
• ISO 14001 (Environmental) & ISO 45001 (Safety) certified
• Zero liquid discharge certified by Gujarat Pollution Control Board
• No ESG-related penalties or controversies
Material ESG Risks – All Mitigated to LOW/VERY LOW
• Chemical spills: ₹50 Cr insurance, zero incidents in 5 years
• Water scarcity: 95% water recycling, rainwater harvesting
• Carbon tax exposure: Renewable investments, pricing pass-through
• Labor strikes: Above-market wages, ESOP, grievance redressal
SECTION 21: COMPREHENSIVE RISK ANALYSIS
Overall Risk Assessment: MODERATE-HIGH (6.5/10)
14 Key Risks Identified & Assessed
VERY HIGH RISKS (8-10/10):
- Valuation Risk (8.5/10): 54x P/E leaves little room for error; 70% probability of near-term correction
- Battery Materials Execution (8.0/10): Unproven vertical; ₹300-400 Cr capex at risk; 60% probability of underperformance
HIGH RISKS (6-8/10):
- Working Capital Elevation (7.5/10): 195 days (vs 150 optimal); ₹60-80 Cr cash flow drag
- China Supply Dependence (6.0/10): 40% raw materials from China; geopolitical/export restriction risk
- Customer Concentration (4.0/10): Top 10 customers 42% of revenue
MODERATE RISKS (4-6/10):
6 risks including NSS integration, currency volatility, generic pricing pressure, FDA delays, JRS competition, geopolitical tensions
LOW RISKS (2-4/10):
3 risks including Nandesari delays, technology obsolescence, regulatory actions
Risk-Adjusted Return Analysis
Scenario | Probability | 3-Year Return | Risk-Adjusted Contribution
Bull Case | 25% | +45% CAGR (₹661 → ₹1,400) | +11.25%
Base Case | 50% | +18% CAGR (₹661 → ₹980) | +9.00%
Bear Case | 25% | -8% CAGR (₹661 → ₹550) | -2.00%
Expected Risk-Adjusted Return: +18.25% per annum
FINAL INVESTMENT RECOMMENDATION
Rating: BUY (4 out of 5 stars)
Target Price: ₹980-1,050 (12-18 months) | ₹1,400-1,600 (3-5 years)
Upside Potential: +48% to +59% (12-18 months) | +112% to +142% (3-5 years)
Risk Profile: MODERATE-HIGH
Investment Horizon: 3-5 YEARS (Long-term wealth creation)
Weighted Overall Score: 7.94/10
Dimension | Score | Weight | Weighted Score
Business Quality | 9.0/10 | 15% | 1.35
Competitive Moats | 8.1/10 | 15% | 1.22
Management & Governance | 8.3/10 | 10% | 0.83
Financial Health | 8.4/10 | 15% | 1.26
Growth Prospects | 8.5/10 | 20% | 1.70
Valuation | 5.5/10 | 15% | 0.83
ESG Profile | 8.1/10 | 5% | 0.41
Risk Management | 6.8/10 | 5% | 0.34
TOTAL | – | 100% | 7.94
INVESTMENT THESIS – WHY BUY
• Industry-leading margins (39.7% EBITDA, 27.6% PAT) – 15-20 points above peers
• Proven execution: 49% PAT CAGR (FY23-25); IPO, NSS, Nandesari on-track
• Capacity doubling: Nandesari adds 70% capacity (51,200 MT) by Q4 FY26
• Strategic NSS acquisition: Europe footprint + infant nutrition + 65% unutilized capacity
• Battery materials optionality: ₹800-1,000 Cr revenue by FY30 (free call option)
• Regulatory moats: 36 certifications create 10-12 year entry barriers
• Fortress balance sheet: 0.10x net debt/equity; ₹88 Cr IPO proceeds for growth
• Customer stickiness: 83.6% repeat business; 7+ year relationships
WHY CAUTION
• Premium valuation (54x P/E) – requires flawless execution
• Working capital elevated (195 days) – cash flow normalization risk
• Multiple initiatives strain management bandwidth
• Battery materials unproven – 60% probability of underperformance
• Small-cap liquidity (4.6% public float) – volatility risk
• China dependence (40% raw materials) – geopolitical/supply chain risk

INVESTOR SUITABILITY
Investor Profile | Suitability
Growth Investors | Highly Suitable
Value Investors | Moderately Suitable (premium valuation)
Conservative Investors | Low Suitability (volatility risk)
Momentum Traders | Low Suitability (low liquidity)
ESG-Focused Investors | Suitable (8.1/10 ESG score)
Dividend Seekers | Not Suitable (no dividend policy)
RECOMMENDED INVESTMENT STRATEGIES
For AGGRESSIVE Investors (High Risk-Reward):
• Entry: ₹650-680
• Position: 5-7% of equity portfolio
• Approach: Lump sum
• Target: Hold 5+ years, exit above ₹1,500
• Stop Loss: ₹520
For MODERATE Investors (Balanced):
• Entry: ₹600-650
• Position: 3-5% of equity portfolio
• Approach: SIP over 3-6 months
• Target: Partial exit at ₹1,000; hold balance 3-5 years
• Stop Loss: ₹550
For CONSERVATIVE Investors (Risk-Averse):
• Entry: ₹550-600
• Position: 2-3% of equity portfolio
• Approach: SIP over 6-12 months
• Target: Exit 50% at ₹900; hold 50% for 5+ years
• Stop Loss: ₹580
FINAL VERDICT
For your Oorjita FinAI Services subscribers:
Sudeep Pharma is a high-quality, growth-oriented specialty pharmaceutical ingredients company trading at premium valuations that are justified by exceptional profitability (39.7% EBITDA margin), strong regulatory moats (36 certifications), and multiple growth levers (Nandesari, NSS, Battery Materials).
Recommendation: BUY for 3-5 year wealth creation
Target Price: ₹980-1,050 (12-18 months) | ₹1,400-1,600 (3-5 years)
Ideal For: Growth investors, quality-focused portfolios, specialty chemicals thematic investors
Risk-Adjusted Expected Return: +18% per annum
Key Insight: The combination of regulatory barriers (10-12 year approval cycles), best-in-class profitability, and 70% capacity expansion creates a rare opportunity in India's specialty ingredients space. Premium valuation is the cost of owning a market leader – justified for long-term compounding.

DISCLAIMER
This analysis is for educational purposes only. Markets are subject to risks and uncertainties. Please consult your financial advisor before making investment decisions. Past performance is not indicative of future results.
Oorjita FinAI Services
Investing Beyond Today
Website: oorjita.ai
Newsletter Editions:
• Morning Brief (Pre-Market Analysis) – Daily 8:00-8:30 AM IST
• Evening Update ("What We Missed") – Daily 7:00-7:30 PM IST
• Weekly Market Manthan – Every Sunday
• Quarterly Company Deep-Dives (Samiksa Oorjita Series)
• Hidden Gems Analysis
COMPLIANCE & DISCLAIMER
www.oorjita.ai is not operated by a broker, a dealer, or a registered investment adviser. Under no circumstances does any information posted on www.oorjita.ai represent a recommendation to buy or sell a security. The information on this site, and in its related newsletters, is not intended to be, nor does it constitute investment advice or recommendations. The information on this site is in no way guaranteed for completeness, accuracy or in any other way. In no event shall Oorjita Fin AI Services be liable to any member, guest or third party for any damages of any kind arising out of the use of any content or other material published or available on www.oorjita.ai, or relating to the use of, or inability to use, www.oorjita.ai or any content, including, without limitation, any investment losses, lost profits, lost opportunity, special, incidental, indirect, consequential or punitive damages. Past performance is a poor indicator of future performance.
Oorjita FinAI Services | www.oorjita.ai | insights@oorjita.ai
Subscribe to unlock premium content
Independent research, deep company analysis, and quarterly insights -
designed to help you think clearly, not trade noisily.
DHARMAJ CROP GUARD LIMITED

NATCO PHARMA LIMITED

10 Productivity tools that are worth checking out

DHARMAJ CROP GUARD LIMITED

NATCO PHARMA LIMITED

DHARMAJ CROP GUARD LIMITED











